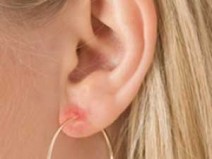
 Do your ears swell up when you wear certain types of jewelry? You might have metal hypersensitivity and these fall into several categories and sources:
Do your ears swell up when you wear certain types of jewelry? You might have metal hypersensitivity and these fall into several categories and sources:
Metal pollution
Then there is the matter of hypersensitivity to metals such as nickel and chromates. These are cell-mediated immunity reactions, without antibody involvement. These too are probably best not called allergies, but clearly there is a sensitization effect that fulfils the prescription of the von Pirquet phenomenon. Don’t forget too that metals can be poisons as well as sensitizers. Mercury and lead are pretty obvious poisons, but so are aluminium (from cooking), cadmium (from tobacco), thallium (rat poisoning and industrial exposure), zinc and iron smelting wastes are EPA regulated toxic materials yet are sold and used legally as fertilizers. There are many other potential metal poisons and it has been my contention for some years now that what I call “metal pollution” may turn out to be a far more serious hazard than chemical pollution, such as pesticides and atmospheric pollution.
Metal Overload
Some people are plagued by reactions to metals with which they come in contact. It can mean they are unable to wear certain types of jewellery, or may find themselves unable to handle money.
These are usually Type IV hypersensitivity (53) reactions, especially nickel and chromate allergy. Delayed dermal hypersensitivity testing (patch test) can generally pinpoint this kind of allergy. Sensitivity, once established, is lifelong.
NICKEL
One in every ten women is allergic to nickel. Almost all cases are women who have had their ears pierced. The problem may begin on the earlobes but soon spreads elsewhere, wherever the skin comes in contact with watches, buckles, clips, etc.
Nickel is widespread throughout the environment and it is almost impossible to avoid contact with it completely.
Sources of Nickel Contact
- Jewellery: Earrings or clasps, necklaces or chains, bracelets, rings, watchbands and buckles.
- Metal fasteners on clothing: Suspenders, hooks and eyes, snap fasteners, zips, buckles, eyelets on shoes, safety pins, buttons, (some of these can be replaced by nylon or rubber equivalents)
- Personal article: Keys, lighters, pocket knives, lipstick holders, eyelash curlers, hairpins, curlers, hair slides, spectacle frame, umbrella handles, handbag fasteners
- Around the home: Needles, pins, thimbles, scissors, paper clips, pens, door handles, cutlery, vacuum cleaners, sewing machines, telephone dials, metal chains, nails and screws, prams, metallic hair dyes. Some detergents contain small amounts of nickel
- Money: ‘Silver’ coins
- At work: metal alloys used in manufacturing, electronics parts and circuits, insecticides and fungicides, storage batteries, nickel catalysts, mordants (used in textile dyeing), hair dyes, electroplating, inks, spark plugs, duplicating fluids, ceramics
If contact with an object is unavoidable it can be painted with clear varnish or covered with tape. Ear-ring clasps may be painted temporarily with nail varnish for limited use. Generally, the effects of perspiration make reactions worse, therefore nickel allergy is worse in summer.
OTHER METALS
Stainless steel is usually non-allergic. It may contain nickel, but it is so strongly bound that even sweat fails to dissolve it.
Gold is far less apt to cause allergy than other decorative metals., but even ‘pure’ (24-carat) gold may be contaminated with traces of nickel or other metals. Sulphur and other chemicals in smog can tarnish gold, which may then cause a reaction
ALUMINIUM
OUR bodies slowly accumulate a toxic load of aluminium from food, water and medicines. Aluminium may well be a major cause of premature senile dementia (Alzheimer’s disease), now epidemic in Britain. Certainly kidney dialysis patients can experience irreversible brain damage if they accumulate too much aluminium. the facts so far suggest that aluminium accelerates the ageing process in normal men and women by the very fact that it accumulates slowly over decades rather than acting as an acute (immediate) poison.
European ‘safe’ limits have been set at 200 microgrammes of aluminium per litre of drinking water. This limit is not based on any scientific facts, it is entirely arbitrary. Significantly, water in many areas of Britain exceeds even this limit, some water having over 300 microgrammes per litre at times.
Aluminium compounds generally are added to beer to improve its clarity and to cheese during processing. Aluminium is an ingredient in baking powders used to make biscuits, cakes and breads. Aluminium compounds are also added to such foods as frozen strawberries, maraschino cherries and pickles – primarily to improve their appearance.
In the United Kingdom about 16,000 tons of aluminium sulphate are added to water supplies every year as a clarifying agent. Aluminium in tap water can be increased by ten or a hundred times by boiling the water in an aluminium saucepan, and can reach twenty times the European safe limit or more if the water is acidified with vinegar or the natural acids of fruit or vegetables.
Avoid aluminium cooking utensils and “tin foil” (which is aluminium).
Connect with us: Facebook – Twitter – LinkedIn – Google+ and take a look at our videos!
The post Hypersensitivity to Metals appeared first on Alternative Doctor Dev Site.
